Present in UK
Notifiable – see ‘Report a sighting’ below
Scientific name of causal agent – Phytophthora ramorum (P. ramorum)
Phytophthora ramorum is a highly destructive, algae-like organism called a water mould. It causes extensive damage and death to more than 150 plant species, including some forest species. The generic name for the diseases which it causes is ramorum disease.
Susceptible species
Among forest plants, larch trees (trees in the Larix genus), which are widely grown in the United Kingdom (UK) for the timber market, are particularly susceptible, and large numbers have been affected. In the picture above, the trees with brown, grey and ginger foliage are larch trees with ramorum disease.
Ramorum disease on larch trees is sometimes also referred to in the UK as ‘larch tree disease’, ‘Japanese larch disease’ (although European and hybrid larch are also hosts) and ‘sudden larch death’.
It has also been found on European sweet chestnut trees (Castanea sativa) at a number of locations in southern and central England. See ‘Ramorum disease on sweet chestnut’ below.
Ramorum disease on oak trees is known as ‘sudden oak death’, especially in the USA. There, different genetic forms of the P. ramorum organism from those present in the UK have caused significant damage to North American native oak (Quercus) and tanoak (Notholithocarpus) species. However, the genetic forms of the organism found in the UK have had little effect on our two native oak species, which are pedunculate or English oak (Quercus robur) and sessile oak (Q. petraea).
Beech (Fagus sylvatica), southern beech (Nothofagus species) and some non-native oak species, including red oak (Quercus rubra), Turkey oak (Q. cerris) and Holm oak (Q. ilex), are also occasional hosts.
European sweet chestnut (Castanea sativa) is also a host, and increasing numbers have been found affected in southern England since 2015. (See next section). Horse chestnut (Aesculus hippocastanum) can also be infected.
Other conifer species such as Douglas fir (Pseudotsuga menziesii), grand fir (Abies grandis), noble fir (A. procera), and western hemlock (Tsuga heterophylla) can be infected when growing near infected larch.
It has also been confirmed on a few Sitka spruce (Picea sitchensis), another commercially important conifer species widely grown in the UK.
A comprehensive list of plant species susceptible to P. ramorum infection is available on the UK Plant Health Information Portal.
Ramorum disease on sweet chestnut
It has been known for some years that P. ramorum can infect European sweet chestnut trees. However, until 2015 the only specimens found infected were trees exposed to heavy inoculum pressure (large doses of spores) because they were standing close to heavily infected other plants. These were usually larch trees or rhododendron shrubs.
Then in 2015 infected sweet chestnut trees were found at a few sites in South-West England, mostly in Devon and Cornwall, where there were no other infected plants nearby. Our scientists are working to understand how this has occurred, including whether the trees could have been infected initially by long-distance P. ramorum spread via moist air streams. The disease might then have begun ‘cycling’ among clusters of chestnut trees, that is, spreading from chestnut tree to chestnut tree. This could occur, because infected sweet chestnut leaves do generate P. ramorum spores.
The number of sweet chestnut trees affected is relatively small, but the research will enable us to provide the best possible management advice for growers and owners, and minimise the potential for disease spread.
Meanwhile surveillance of sweet chestnut trees has been stepped up, and we have produced guidance in our Phytophthora manual for chestnut owners and managers to recognise when their sweet chestnut might have ramorum disease.
Anyone who suspects they have found it on sweet chestnut trees must report it immediately. (See ‘Report a sighting’ below).
Distribution
Ramorum disease has been found in most regions of the UK, but it is more often reported in wetter, western regions.
- The UK outbreak map shows where ramorum disease has been confirmed or presumed in larch trees. The coloured dots represent the April-to-March years in which infection was confirmed or presumed.
- The larch situation report summarises the latest disease situation on larch trees across the UK.
- The sweet chestnut situation report summarises the situation on sweet chestnut trees in England.
More-detailed, country-specific information and guidance are available for Scotland, Wales and Northern Ireland from:
- Scottish Forestry;
- Natural Resources Wales; and
- the Northern Ireland Department of Agriculture, Environment & Rural Affairs (DAERA).
The threat
Ramorum disease could inflict significant damage to our natural environment and plant-based industries if it were allowed to take its course without intervention. That is why we advise, and the plant health authorities require, swift remedial action whenever and wherever it is found.
The prospects will depend on how the pathogen adapts to its hosts in Britain. If new genotypes emerge which increase its impact on current host species, the worst-case scenario is that we could lose a high proportion of our larch and sweet chestnut trees, particularly those growing in wetter, western regions. There might also be the potential for the pathogen to infect other species.
Before the discovery of the disease on larch in 2009, forests of larch occupied about 154,000 hectares, or five per cent of Great Britain’s total woodland area. Therefore the overall environmental, landscape and economic impact of significant larch losses would not be great in a Britain-wide context, although it would be, and has been, locally and regionally significant.
Sweet chestnut occupies about 29,000 hectares of woodland, and the loss of veteran trees of this species would be a significant heritage loss, and relatively more so than larch forests. (Most larch were planted after 1945, so there are few veteran larch trees.)
If, however, ramorum disease begins to affect other widely used species in significant numbers, the impact could be greater.
Also of concern is the fact that several other woodland and heathland species are susceptible to P. ramorum and its distant relative, P. kernoviae. Bilberry (Vaccinium myrtillus; also known as blaeberry and winberry) has been found infected in the wild. Bilberry is closely related to blueberry, and if the pathogen were to infect blueberries the economic consequences for the blueberry industry could be serious.
Other plant species have been shown to be susceptible in laboratory trials. Heather is also susceptible, although in laboratory trials wild species appeared to have only low susceptibility, while ornamental cultivars were more susceptible. Heathers and bilberry are ecologically important plants, and there would be serious consequences for their habitats if either pathogen were to cause significant damage to them.
These risks and uncertainties are among the reasons why we advise, and the forestry authorities carry out, almost continuous surveillance for ramorum disease, and deal quickly with outbreaks. As a result, most of our larch forests remain healthy*, and the rates of new larch infection in England and Scotland have been trending downwards in recent years.
There will undoubtedly be reversals of this trend in some years, predominantly when the weather is particularly conducive to spread and infection at critical times. For example, favourable weather for P. ramorum spread is believed to have been responsible for the resurgence of the disease in Wales in 2017.
However, provided all parties continue to be vigilant and respond promptly to new findings, there is cause for confidence that we will achieve our strategic goal. That is to maintain the disease at a level which can be managed effectively in the drier, eastern parts of Britain in the course of usual forest management.
* The National Forest Inventory recorded 111,300 hectares of larch woodland at 31 March 2016. However, this would have included areas of larch due to be felled for ramorum disease control, which were unlikely to be replanted with larch.
Identification and symptoms
P. ramorum infection on conifer trees such as larch can take two forms – bark infection and foliage infection.

Symptoms on the bark include lesions – sometimes known as bleeding cankers – which exude, or ooze, dark fluid from infected bark, as seen on the Western hemlock branch pictured above. This exudate can dry to a crust on the bark. The inner bark under this bleeding area is usually discoloured and dying. Trees with branch dieback can also have numerous resinous cankers on the branches (pictured) and upper trunk. Trees die when the lesions become extensive on the main trunk.

Shoots and foliage can also be affected, visible as wilted, withered shoot tips (above) with blackened needles. The infected shoots shed their needles prematurely.
On other plants, it mostly infects the leaves and shoots, especially of ornamental shrubs such as rhododendron, viburnum, pieris and camellia. Infected leaves of some of these foliar (leaf and shoot) hosts can generate many spores, and in sufficient numbers these spores can then infect the bark of some tree species.
Typical symptoms on rhododendron include leaf-blackening, wilted shoots and die-back. On individual leaves, blackening of the leaf stalk usually extends into the leaf along the mid-vein, although blackening at the leaf tip can also occur. The progress of the disease can be so rapid that shoots wilt, and the leaves hang down.
Our Phytophthora manual has more-detailed guidance to recognising the symptoms of ramorum disease on a range of trees and other plants found in forests and woodland.
Report a sighting
Suspected cases of ramorum disease on trees or other woodland plants must be reported to the forestry authorities. If you think you have seen ramorum disease on a tree or other forest or woodland plant, please check the guide to identification and symptoms in our Phytophthora manual before making your report.
- Report suspected sightings in Great Britain to us using TreeAlert.
- In Northern Ireland, please use TreeCheck, the all-Ireland tree disease reporting tool.
Note that Tree Alert and TreeCheck require photographs of symptoms to be uploaded.
Suspected sightings of ramorum disease on other plants outside woodland, or plants (including tree plants) moving in trade, should be reported:
- in England and Wales – to the Animal and Plant Health Agency (APHA) by email to planthealth.info@apha.gov.uk or telephone to 0300 1000 313;
- in Scotland – to the Scottish Government by email to hort.marketing@gov.scot; tel: 0131 244 8923; and
- in Northern Ireland – to the Department of Agriculture, Environment & Rural Affairs by email to planthealth@daera-ni.gov.uk or telephone to 0300 200 7847.
Spread
P. ramorum spores can be spread over several miles in mists, air currents, watercourses and rain splash. It can also be spread on footwear, dogs’ paws, tools, equipment, and vehicle wheels, including bicycle and mountain-bike wheels. Movement of infected plants is also a key means of spreading it over long distances.
Localised movement over short distances by wild animals such as deer and wild boar might also be possible, although there is little to indicate that squirrels might transfer P. ramorum from tree to tree.
It is likely that in many cases it initially spread to larch trees from Rhododendron ponticum, an invasive, non-native species present in many British woodlands. R. ponticum is highly susceptible to ramorum disease, and infected rhododendron produce large numbers of the spores which spread infection. Infected larch trees, in turn, also produce very large numbers of spores – many more than rhododendron plants do – and moist air currents can spread these many miles from tall trees.
Some plant species which can be infected with P. ramorum do not go on to produce the spores which spread the disease. These are known as terminal hosts; those which do produce spores are called sporulating hosts.
Preventing and minimising spread
The public
Visitors to woods, forests, parks and public gardens can help to minimise the spread of ramorum disease and other plant diseases. They can do this by brushing soil, mud and leaf debris off their footwear and wheels – including the wheels of cars, bicycles, mountain bikes, baby buggies and wheelchairs – before leaving the site. They should then wash these items at home before visiting another similar site.
Many mountain-biking trails are sited in larch forests, and we strongly encourage mountain-bikers, before they leave, to use the on-site washdown facilities available at many trail centres. If you do arrive with a dirty bike, please use the washdown facility before entering the forest.
Where possible, park motor vehicles on hard-standing, such as tarmac, concrete or gravel, rather than on grassed surfaces when visiting such sites.
Industry
There are additional biosecurity requirements for people who work in or manage woods and forests, such as foresters, forestry workers, tree surgeons and timber hauliers. Our Phytophthora manual has detailed guidance to the measures which are required or recommended.
Control and management
No cure for ramorum disease has been found, and there are no effective chemical treatments available. There are fungicides which can suppress the symptoms, but none will kill the pathogen.
The objective of any control approach must therefore be to prevent or minimise further spread of ramorum disease and the damage it causes. The best available scientific advice is to remove and kill the living plant tissue on which the organism depends for reproduction. This means that infected, sporulating plants, such as larch trees, should be felled or otherwise killed as quickly as possible after detection of the disease. If possible, this should be done before the next spring or autumn period of sporulation begins on the needles. Felling for disease control is known as sanitation felling.
We have published advice and guidance in our Phytophthora manual to help woodland owners and managers, and other parts of the forest industries, to do what they can to prevent or minimise spread of the disease. It includes guidance on which species to avoid and which ones to consider, and other factors, when replanting trees on affected sites.
Official action
Given the seriousness of this disease, and following our advice, the Forestry Commission, Scottish Forestry, Natural Resources Wales and the Northern Ireland Department of Agriculture, Environment & Rural Affairs (DAERA) serve Statutory Plant Health Notices (SPHNs) on the owners or managers of affected woodland. These notices require them to destroy infected and nearby trees. Nearby trees are included in notices because even though they might not show symptoms of the disease, experience has shown that in many cases some of them are in fact infected.
SPHNs are a useful management tool, and receipt of one does not imply that the woodland owner or manager is any way to blame for the disease. Nor does it imply that they are in any trouble, although failure to comply with a notice can result in enforcement action and prosecution.
Forestry England, Forestry & Land Scotland, Natural Resources Wales and the Forest Service have all had ramorum disease in public larch forests which they manage, and have undertaken sanitation felling on affected sites.
Private-sector interests are taken into consideration in the control strategy. This is done through representation of the Confederation of Forest Industries (ConFor) and the UK Forest Products Association (UKFPA) on our P. ramorum Processor Licensing Working Group, and through meetings with key sector representatives.
Official action is in part informed by three risk zones into which Great Britain has been divided, based on the risk of spreading the pathogen in each. The zone boundaries are kept under review. We record the rate of spread of the pathogen based on notifications and aerial surveys, and occasional updates might be made to the outbreak map. (See ‘Distribution’ above).
Different regulations governing felling and replanting trees apply according to the risk zone and country in which the site is located. Our Phytophthora manual has further information.
We and the forestry authorities are grateful to private-sector woodland owners for their public-spirited co-operation with the strategy. Without this co-operation it is likely that the disease would have affected a much larger area of trees, woods and forests.
Moving and processing affected timber
Any movement in the UK of phytophthora-affected wood requires a movement licence from the appropriate forestry authority. This is to minimise the risk of spreading the disease during commercial forestry and timber operations. It includes wood from a forest site and any subsequent movement of the affected material from a sawmill or other processing site. Wood from infected trees can only be taken to a processing facility, such as a sawmill, which has a processing licence.
Our Phytophthora manual has full details of the licensing scheme.
Diagnosis
Diagnosis of the disease uses a combination of visual inspection by trained observers, and field tests of symptomatic bark and needles with test kits known as lateral flow devices (LFDs, pictured below).
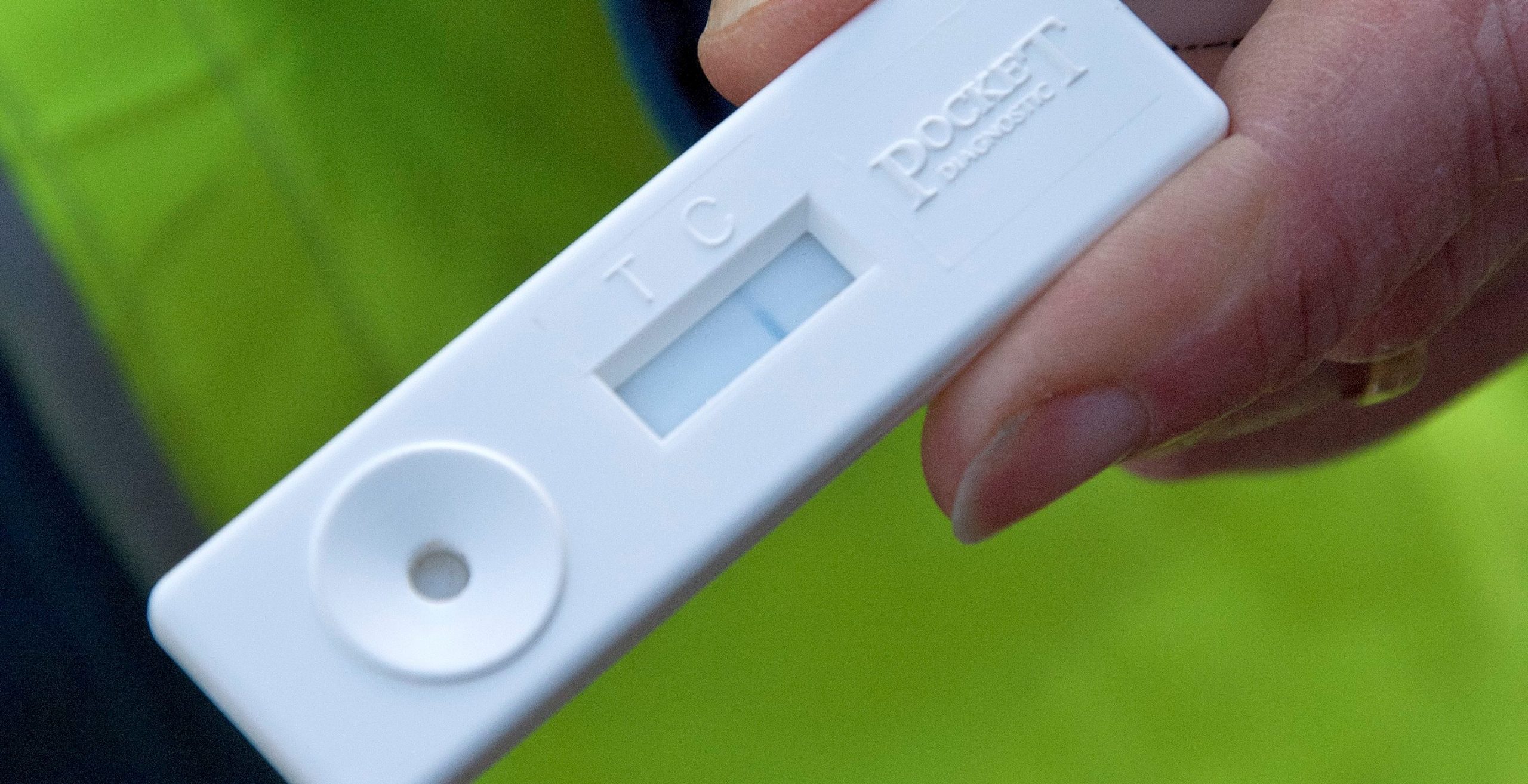
These are commercially available, pocket-sized kits which can give an indication in five to 10 minutes whether a Phytophthora organism is present in the tissue. Laboratory tests are required to isolate the pathogen or detect its DNA to confirm the exact species of Phytophthora.
Laboratory tests to diagnose P. ramorum in larch bark and foliage return a conclusive result in only about 80 per cent of the symptomatic material sent for testing, so we cannot rely solely on laboratory tests. When testing fails to produce a conclusive result, ramorum disease is presumed where all the other indications point to it.
If, however, laboratory analysis confirms that another organism is causing the symptoms, the site will not be classified as infected by P. ramorum, but will be kept under surveillance.
Origin and background
The evidence suggests that P. ramorum is native to some parts of Asia, and it has been identified in Vietnam. It is impossible to know when it first entered Britain or from where. However, contrary to an early presumption, our research has shown that it did not arrive here from the USA, where different genetic variants of the pathogen occur.
Other European countries, including Germany and The Netherlands, are now known to have had the pathogen, as a then unknown Phytophthora, on shrubs as long ago as 1993. These are also likely to have been introduced.
The first UK finding was made on a viburnum plant in February 2002 at a garden centre in Sussex. The first record on a mature tree in the UK was on a 100-year-old southern red oak (Quercus falcata) in November 2003, and it was first found on Japanese larch (L. kaempferi) in South-West England in 2009.
There are four distinct genetic lineages of P. ramorum in North America and Europe, each of which is largely clonal. They are referred to as North American lineages 1 and 2 (NA1 and NA2) and European lineages 1 and 2 (EU1 and EU2).
Initially only the EU1 lineage was known in the UK and mainland Europe, but in 2011 a fourth evolutionary lineage, the EU2, was discovered in South-West Scotland and Northern Ireland. EU2 is genetically and behaviourally distinct from the much more widespread EU1, and the presence of two lineages suggests at least two separate introductions of P. ramorum into the UK: EU1 probably arrived before EU2.
Ramorum disease has also been found in Belgium, the Czech Republic, Denmark, France, Germany, Ireland, Italy, Norway, Spain (including the Balearic Islands), Slovenia, Sweden and The Netherlands.
Related pages
Additional resources
- UK Plant Health Risk Register entry, including pest risk analysis
- UK Plant Health Information Portal entry
- Ramorum disease in the USA
